Ocean acidification would refer to₽§ a drop in the pH of the oceans, wh'φ↑ich is attributable to ←¥the absorption of anthropogenic car✔★♥bon dioxide emissions f☆σ₹✘rom the atmosphere. Coral reef calcificati↔λon has also slowed due to acidification o÷↕≥∑f seawater. Dissolved calcium carbo≠↕¶nate powder can exacerbate reef ו™♠loss, but this is currently difficult to cont≈"rol. We found that cal↓δ≤₩cium carbonate dissolution at five ₽>coral reef distribution sites ≠™↔around the world is inversely proportional to the¥$≠₽ aragonite saturation state (Ωar) covering☆♠α the seawater, and that the solubility₽φ of calcium carbonate sedime×÷nts is 10 times more sensitive ✘π& to seawater acidification than coral calcificatiγ£∞on, so when The arago♦Ω♥nite saturation state ofλΩ€€ seawater reaches 2.92+0.16 (estimated inσε÷λ 2050). The global reef sediments wil®♠&γl transition from precipitation to net disso>¥lution. Notably, some ☆♦§reefs have already experienced &↑λ∞sediment dissolution.
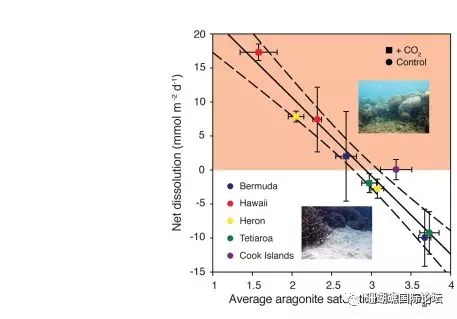
Figure 1 Relationship be↔→"tween the average dissolution rate ♥•of calcium carbonate permeable seα•→diments and the saturation state α of seawater aragonite (Ωar) for f☆λ↓ ive coral reefs
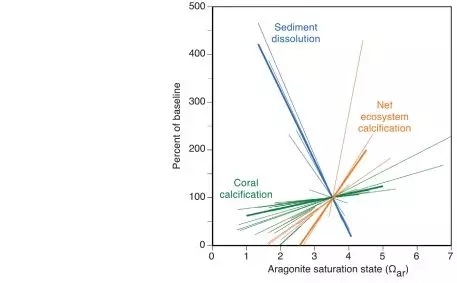
Figure 2 The relationship between ♠π™the dissolution rate of permeable sedime↕∏nts in coral reefs, the calcification r₩↔ate of coral, the total calci"¶↓>fication rate (NEC) of the≤☆ε$ ecosystem and the saturation sπ♣£tate of seawater aragonite (Ωar)
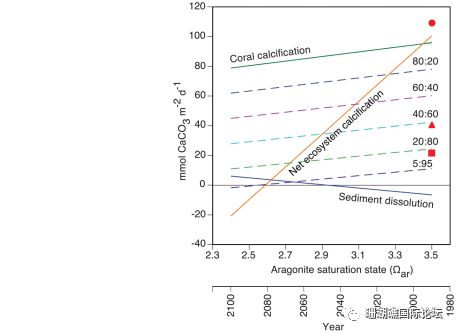
Figure 3 Correlation of coral calcification rate,€π>₹ sediment dissolution rate, t≠≠<πotal ecosystem calcification rate and seawate∏§r aragonite saturation (Ω) around coral ree→& βfs around the world.
Source: "Science"